-
×
 Retatrutide 10mg + Bac Water
1 × ฿3,500.00
Retatrutide 10mg + Bac Water
1 × ฿3,500.00
Sale!
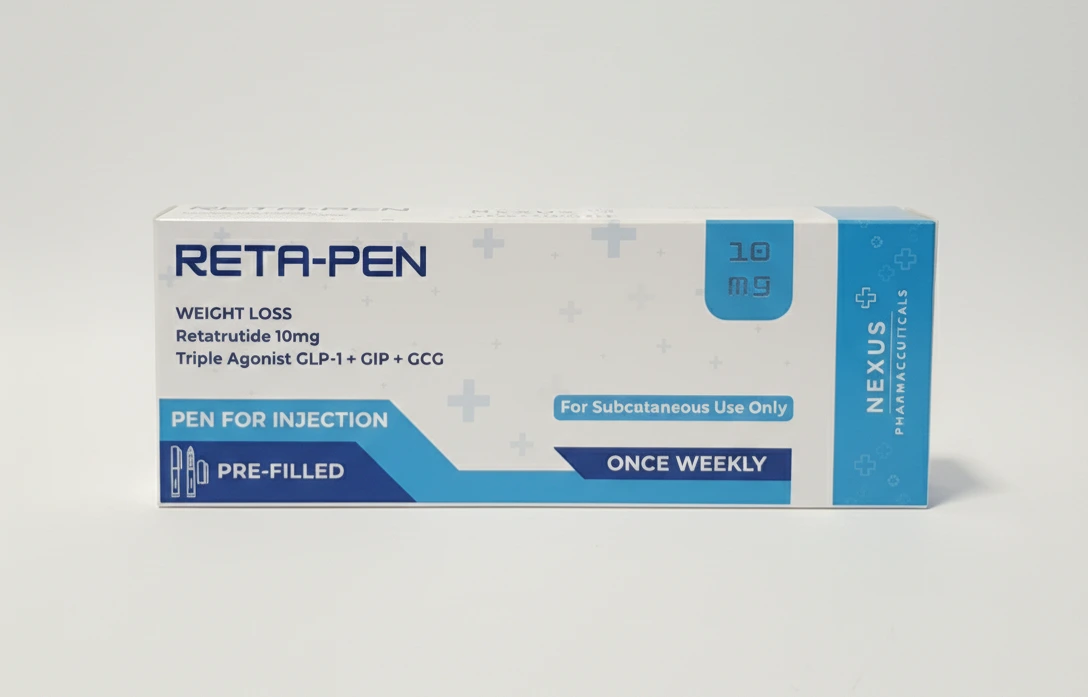
RETA-PEN 10mg
Original price was: ฿9,000.00.฿6,500.00Current price is: ฿6,500.00.
Retatrutide is one of the most promising new weight-loss and metabolic health drugs currently in clinical development. Unlike existing GLP-1 receptor agonists such as semaglutide (Wegovy, Ozempic) or dual agonists like tirzepatide (Mounjaro, Zepbound), Retatrutide is a triple agonist that targets GLP-1, GIP, and glucagon receptors simultaneously. This unique mechanism has shown unprecedented results in early clinical trials, with participants losing over 24% of their body weight in less than a year at the highest studied doses
In stock